Audit-ready release visibility
Track releases, revisions, and approvals with a clear audit trail for QA/RA and Regulatory teams.
EU MDR/IVDR plus FDA ready
Deliver compliant eIFU access across EU and US markets with multilingual distribution built in.
Operationally scalable delivery
Automate publishing and access so teams stop chasing outdated IFUs and manual updates.
Admin and Web App Features: Control, Compliance, and Confidence
Run compliant eIFU workflows with admin tools for governance and a web app that gives users fast, reliable access to current instructions.
Admin Dashboard Features
Most Popular
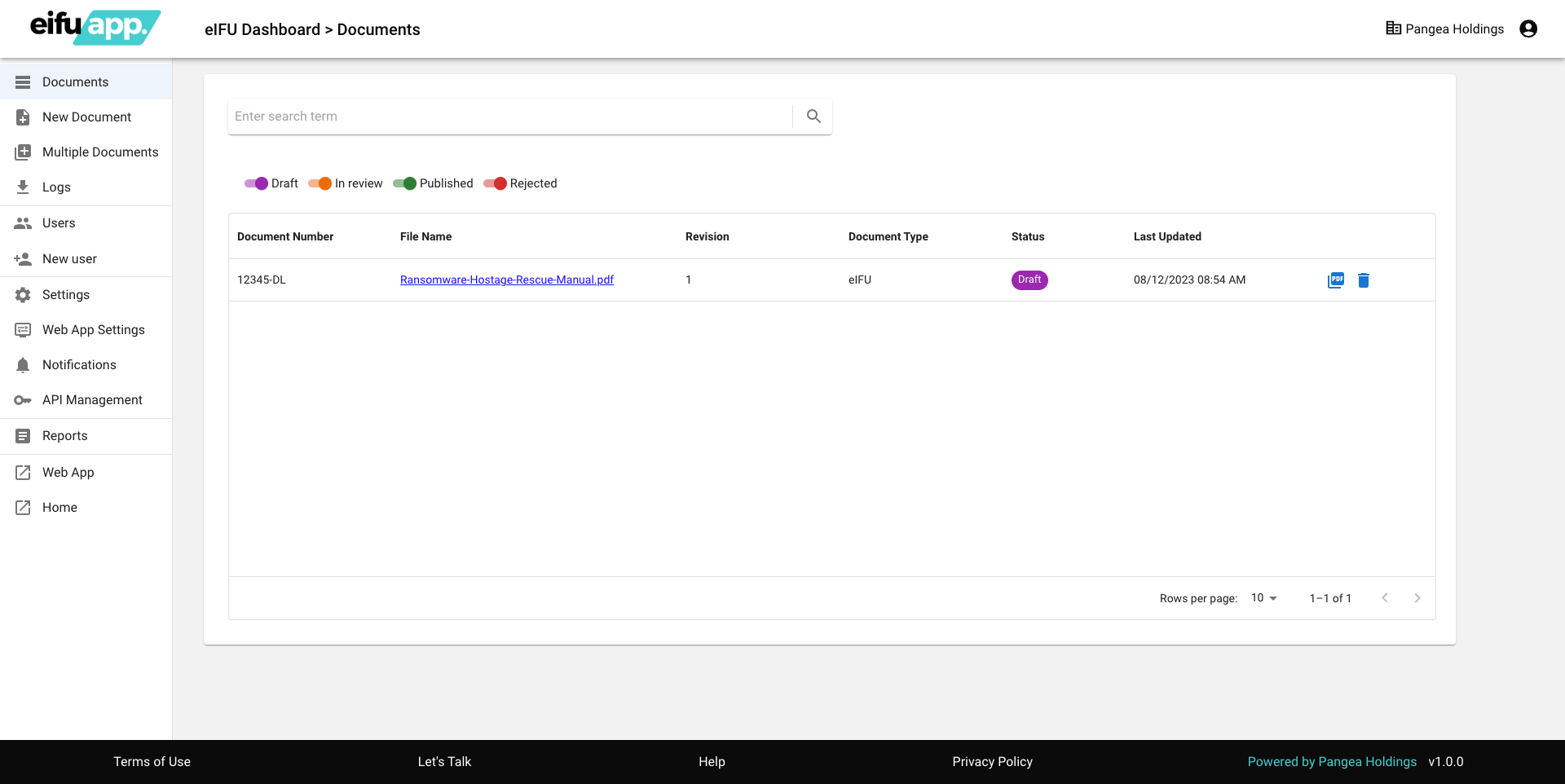
- User Management: Control access efficiently.
- Document Organization: Effortlessly oversee documents.
- Business Unit Streamlining: Manage units seamlessly.
- Bulk Document Upload: Rapidly upload multiple documents.
- Insightful Analytics: Access comprehensive insights.
- Personalized Settings: Customize to preferences.
- URL Redirections: Manage for optimal access.
Web App Features
Most Popular

- Document Search: Find documents effortlessly.
- URL Sharing: Share with unique URLs.
- Details, Revisions, Updates: Access comprehensive document insights.
- Stay Updated: Receive document updates.
- Swift Downloads: Obtain documents swiftly.
- Region & Language Filters: Tailor search preferences.
- Multilang Support: Access docs in multiple languages.
Control releases. Reduce risk. Give every team a clear, compliant path to the latest IFU.
Explore role-specific eIFU pages
Use the page that matches your team's buying intent:
